
All other bonds are somewhere in-between. Very small differences in electronegativity result in non-polar covalent bonds, and very large differences in electronegativity result in ionic bonds. Only the extreme cases are very clear. The attraction between oppositely charged ions is called an ionic bond, and it is one of the main types of chemical bonds in chemistry.Summary of Electronegativity and Bond formation.In this case the atoms with the higher electronegativity will be fully negative (due to the "gaining" of an electron) while the other atom is fully positive (due to its virtual loss of an electron). know that ionic bonding is the strong electrostatic attraction between oppositely charged ions Topic 13: Enegetics II.If for some reason the bond between the atoms is broken, the atom with the higher electronegativity will actually keep the electron for itself. Ionic bonds result from the electrostatic attraction between oppositely charged ions, which form when valence electrons are transferred from one atom to another. The coordinate compounds are little more complicated ionic compounds. 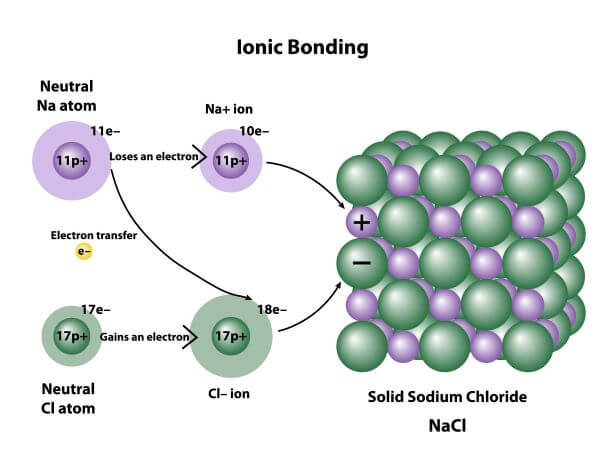
The total charge on the compound is zero as positive and negative charges balance each other. Ionic bonds can occur between metals and. Electronegativity and Ionic Bonding An Ionic bond is the bond formed by the complete transfer of valence electron to attain stability. Define the following terms: a) ionic bond b) cation. An ionic bond is the electrostatic force of attraction between two oppositely charged ions (called anions and cations). An ionic bond is a bond between ions where oppositely. Coordinate bonds may also be referred to as Dative bonds or dipolar bonds. individual molecules in ionic compounds, just the regular stacking of positive and negative ions. The opposite charges on the ions cause the ions to bond, or be held together, by electrostatic forces. In other words, the shared pair’s electrons are both from the same atom. It is an alternative covalent bond in which the electron pair is shared by only one atom. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
